robdiaz to further illustrate, look at how the model of the atom has changed so drastically over time...
1. this was John Dalton's model of the atom (basically the same as what Democritus had proposed years ago). it was just considered to be a small, indivisible unit. that's all they knew about it.
2. Through experimentation, JJ Watson "discovered" the electron. he didn't actually see them (we still to this day can't really see electrons) but he knew they existed. he proposed this model which at the time was the best theory and was thus accepted. he basically said there were a few tangible electrons, and just a "soup" of positive charge to make the whole unit neutral.
3. Ernest Rutherford in his famous gold foil experiment was able to demonstrate that the positive charge of an atom was highly concentrated into a center nucleus. This was the first time anybody proposed the idea of a nucleus. So he used Thompson's model and adapted it to incorporate a nucleus.
4. the next big stepping Neils Bohr's model, who by studying emitted photons determined that electrons orbit at varying levels on energy/distance from the nucleus, instead of just a seemingly random swirl like the previous model.
and even this has changed where rather than electrons occupying simple orbitals, its more of an electron cloud at which point an electron can occupy any space of that "cloud."
tl;dr... scientific theory is completely based on evidence, and with introduction of new evidence, the accepted theory will change. speciation and evolution are currently the best working theory we have that explains the diversity of life on earth, and is a result of many pieces of evidence left behind.

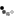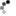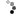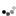
 Reply With Quote
Reply With Quote











